Automotive Cam Chains Market Share Analysis by Leading Companies and Emerging Players
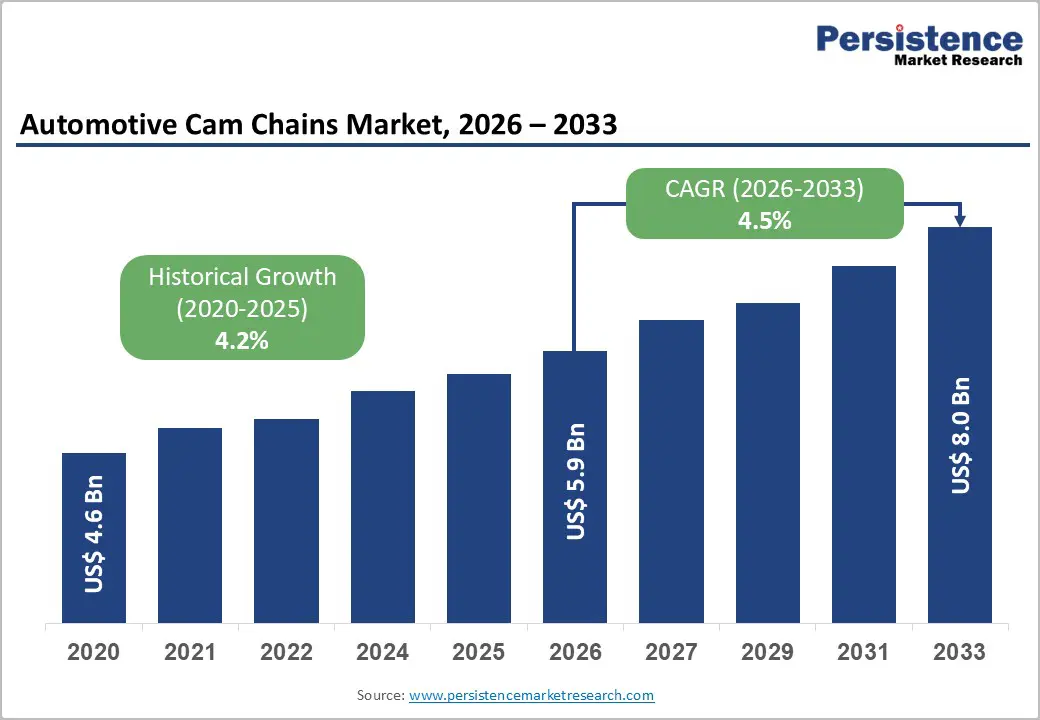
The global automotive cam chains market is witnessing stable and sustained growth, driven by the rising demand for durable and efficient engine components across passenger and commercial vehicles. According to recent insights from leading market research firms, the automotive cam chains market is projected to be valued at US$5.9 billion in 2026 and is expected to reach approximately US$8.0 billion by 2033, expanding at a compound annual growth rate (CAGR) of 4.5% during the forecast period from 2026 to 2033.
Cam chains play a critical role in internal combustion engines by synchronizing the rotation of the crankshaft and camshaft, ensuring precise valve timing for optimal engine performance. Unlike timing belts, cam chains offer higher durability, longer life cycles, and reduced maintenance requirements, making them increasingly preferred by automakers and consumers alike.
The growth of the automotive cam chains market is primarily driven by the increasing production of vehicles globally, particularly in emerging economies. Rising consumer preference for low-maintenance automotive components, coupled with advancements in engine technologies, has further propelled demand. Additionally, the continued relevance of hybrid vehicles and internal combustion engine (ICE) platforms in the transition toward electrification supports the steady adoption of cam chains.
Get the full report here:
https://www.persistencemarketresearch.com/market-research/automotive-cam-chains-market.asp
Key Highlights from the Report
➤ The automotive cam chains market is projected to grow from US$5.9 billion in 2026 to US$8.0 billion by 2033, registering a CAGR of 4.5%.
➤ Increasing vehicle production, particularly in emerging markets, continues to drive consistent demand for cam chain systems.
➤ Passenger vehicles dominate the market, owing to higher production volumes and widespread adoption of durable engine components.
➤ Metal-based cam chains remain the preferred type due to their strength, reliability, and long operational lifespan.
➤ Asia-Pacific leads the global market, supported by strong automotive manufacturing ecosystems in countries such as China, India, and Japan.
➤ Technological advancements in chain design, coatings, and lubrication systems are enhancing durability and reducing noise levels.
Automotive Cam Chains Market Share Analysis by Leading Companies and Emerging Players
The global automotive cam chains market is witnessing stable and sustained growth, driven by the rising demand for durable and efficient engine components across passenger and commercial vehicles. According to recent insights from leading market research firms, the automotive cam chains market is projected to be valued at US$5.9 billion in 2026 and is expected to reach approximately US$8.0 billion by 2033, expanding at a compound annual growth rate (CAGR) of 4.5% during the forecast period from 2026 to 2033.
Cam chains play a critical role in internal combustion engines by synchronizing the rotation of the crankshaft and camshaft, ensuring precise valve timing for optimal engine performance. Unlike timing belts, cam chains offer higher durability, longer life cycles, and reduced maintenance requirements, making them increasingly preferred by automakers and consumers alike.
The growth of the automotive cam chains market is primarily driven by the increasing production of vehicles globally, particularly in emerging economies. Rising consumer preference for low-maintenance automotive components, coupled with advancements in engine technologies, has further propelled demand. Additionally, the continued relevance of hybrid vehicles and internal combustion engine (ICE) platforms in the transition toward electrification supports the steady adoption of cam chains.
📌 Get the full report here: https://www.persistencemarketresearch.com/market-research/automotive-cam-chains-market.asp
Key Highlights from the Report
➤ The automotive cam chains market is projected to grow from US$5.9 billion in 2026 to US$8.0 billion by 2033, registering a CAGR of 4.5%.
➤ Increasing vehicle production, particularly in emerging markets, continues to drive consistent demand for cam chain systems.
➤ Passenger vehicles dominate the market, owing to higher production volumes and widespread adoption of durable engine components.
➤ Metal-based cam chains remain the preferred type due to their strength, reliability, and long operational lifespan.
➤ Asia-Pacific leads the global market, supported by strong automotive manufacturing ecosystems in countries such as China, India, and Japan.
➤ Technological advancements in chain design, coatings, and lubrication systems are enhancing durability and reducing noise levels.





-tires-market-2026-2033.webp)