U.S. Cold Chain Packaging Market Outlook with Industry Innovation
U.S. Healthcare Cold Chain Packaging: Growth Driven by Biologics and Precision Logistics
The U.S. healthcare cold chain packaging market is entering a high-growth phase, shaped by the rapid expansion of biologics, vaccines, and advanced therapies. As pharmaceutical innovation becomes increasingly temperature-sensitive, packaging is no longer a passive function—it is a critical component of product integrity, regulatory compliance, and patient safety.
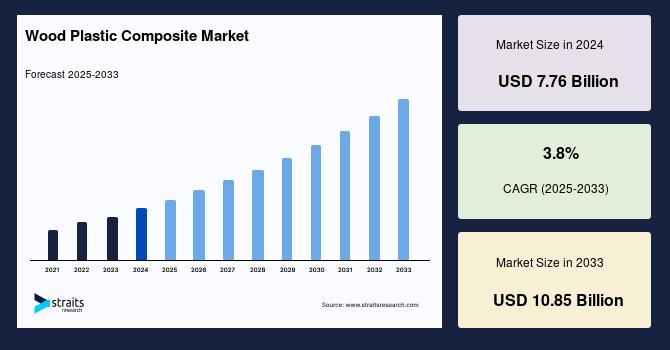
According to a report published by Grand View Research, the U.S. healthcare cold chain packaging market is projected to reach USD 9.32 billion by 2033, growing at a CAGR of 12.2% from 2026 to 2033. This robust trajectory reflects structural shifts in drug development and distribution models, where maintaining precise thermal conditions is essential across increasingly complex supply chains.
The Biologics Boom and Rising Temperature Sensitivity
A central driver of demand is the transition from traditional small-molecule drugs to biologics, including monoclonal antibodies, mRNA vaccines, and cell and gene therapies. These therapies are inherently unstable and require strict temperature control—often within narrow ranges such as 2°C to 8°C or even cryogenic conditions below -80°C.
As a result, cold chain packaging solutions—such as insulated containers, phase change materials (PCMs), and vacuum insulated panels (VIPs)—have become indispensable. Even minor temperature excursions can render high-value drugs ineffective, leading to financial losses and clinical risks. This has elevated packaging from a logistics afterthought to a strategic priority within pharmaceutical operations.
Additionally, the U.S. continues to lead in clinical trials and biopharmaceutical R&D, which further increases demand for reliable packaging systems capable of supporting long-distance and multi-stage transportation.
Technology Integration and Smart Packaging Evolution
One of the most notable trends is the integration of digital technologies into cold chain packaging. Labels and sensors embedded with IoT capabilities now enable real-time temperature monitoring, location tracking, and predictive analytics. These smart systems enhance transparency and reduce risks associated with spoilage or non-compliance.
The labels and sensors segment is expected to grow at one of the fastest rates in the coming years, driven by increasing regulatory scrutiny and the need for end-to-end visibility. Pharmaceutical companies are prioritizing data-driven supply chains, where packaging plays an active role in monitoring and reporting conditions throughout transit.
Moreover, active cold chain packaging—equipped with powered cooling systems—is gaining traction for high-value shipments requiring precise control over extended durations. While passive systems still dominate due to cost efficiency and scalability, active solutions are becoming essential for global and long-haul logistics.
Sustainability and Regulatory Pressures Reshaping Design
Sustainability is emerging as a defining factor in packaging innovation. Companies are increasingly developing recyclable, reusable, and bio-based materials to meet environmental, social, and governance (ESG) goals. This shift is particularly significant in the U.S., where regulatory frameworks and corporate commitments are pushing for reduced environmental impact.
At the same time, compliance with stringent regulations—such as Good Manufacturing Practices (GMP) and Good Distribution Practices (GDP)—is shaping product development. Packaging solutions must undergo rigorous validation to ensure consistent thermal performance, traceability, and reliability.
This regulatory intensity favors established players with proven capabilities, creating high entry barriers and reinforcing long-term supplier relationships within the healthcare ecosystem.
Market Expansion and Strategic Investments
The expansion of biopharmaceutical manufacturing capacity in the U.S. is further accelerating demand for cold chain packaging. Industry investments in supply chain infrastructure—including cold storage facilities and automated distribution centers—highlight the growing importance of temperature-controlled logistics.
Recent industry developments show that pharmaceutical distributors are investing heavily in advanced cold chain capabilities to support the rising volume of specialty drugs. These investments are not only enhancing storage and transportation but also driving innovation in packaging systems that can integrate seamlessly with automated and digitalized supply chains.
In this context, the market’s projected growth reflects both increasing demand and evolving complexity. The U.S. healthcare cold chain packaging market, valued at approximately USD 3.72 billion in 2025, is expected to more than double over the next decade as temperature-sensitive therapies dominate the pharmaceutical pipeline.
Integrated Market Outlook
In essence, the U.S. healthcare cold chain packaging market is transitioning toward a more sophisticated, technology-enabled, and sustainability-driven ecosystem. The projected valuation of USD 9.32 billion by 2033 underscores the critical role packaging plays in modern healthcare delivery.
This growth is not merely volume-driven—it reflects a deeper transformation in how medicines are developed, transported, and administered. As biologics, personalized medicine, and global distribution networks continue to expand, cold chain packaging will remain a cornerstone of pharmaceutical innovation, ensuring that life-saving therapies reach patients safely and effectively.